商品详情
返回产品目录商品包装及说明书因厂家更换频繁,如有不符以实物为主
注射用德曲妥珠单抗
国际零售参考价:¥**/瓶
产品参数 中文说明 外文说明关联产品 本品资讯
-
- 通用名称
- 注射用德曲妥珠单抗
- 商品名
- Enhertu
- 其它名称
- DS-8201、优赫得、T-DXd、曲妥珠单抗\德鲁替康注射剂、fam-trastuzumab deruxtecan-nxki、trastuzumab deruxtecan、恩赫图
- 英文名称
- trastuzumab deruxtecan-nxki
- 包装规格
- 100mg
- 产地及厂家
- 阿斯利康&第一三共制药(AstraZeneca&Daiichi Sankyo)
- 适应症范围
- 2019年12月,HER2阳性乳腺癌晚期后线;
2021年1月, HER2阳性胃癌晚期二线及以后,成为第一个被批准治疗HER2阳性胃癌的ADC;
2022年5月,HER2阳性乳腺癌晚期二线;
2022年8月5日,HER2低表达乳腺癌晚期后线,第一次改写乳腺癌的HER2二分类标准;
2022年8月12日,既往接受过全身治疗的HER2突变不可切除或转移性非小细胞肺癌(NSCLC),成为第一个获FDA批准的用于肺癌的HER2靶向疗法。
- 储存条件
- 冷藏
- 靶点
- HER2
-
温馨提醒:本说明书仅供参考,最新的说明书详见药品附带的说明书。
警告:间质性肺疾病和胚胎安全性毒性
- ENHERTU报道了间质性肺疾病(ILD)和肺炎,包括致命病例。监测并及时调查体征和症状,包括咳嗽,呼吸困难,发烧以及其他新的或恶化的呼吸道症状。在所有2级以上ILD /肺炎患者中永久停用ENHERTU。告知患者风险和需要立即报告症状的信息[请参阅剂量和用法(2.2),警告和注意事项(5.1) ]。
- 胚胎-胎儿毒性:怀孕期间暴露于ENHERTU会导致胚胎-胎儿伤害。告知患者这些风险和有效避孕的必要性[请参阅警告和注意事项(5.4),在特定人群中使用(8.1,8.3)]。
1适应症和用途
ENHERTU适用于治疗无法切除或转移性HER2阳性乳腺癌的成年患者,这些患者已在转移环境中接受了两种或更多种基于抗HER2的先前治疗方案。
该适应症是根据肿瘤缓解率和缓解持续时间在加速批准下批准的[见临床研究(14.1) ]。继续批准该适应症可能要取决于验证性试验中对临床益处的验证和描述。
2剂量和给药
2.1建议剂量和时间表
请勿将ENHERTU替代曲妥珠单抗或ado-曲妥珠单抗emtansine或与其替代。
ENHERTU的推荐剂量为5.4 mg / kg,每3周(21天周期)静脉输注一次,直至疾病进展或出现不可接受的毒性。
第一次输注:在90分钟内进行输注。
后续输注:如果先前的输注耐受性良好,则应进行30分钟以上的给药。
如果患者出现输液相关症状,请减慢或中断输液速度。
如果发生严重的输注反应,请永久停用ENHERTU。
2.2剂量修改
如表1和表2所述,不良反应的处理可能需要暂时中断,降低剂量或中止ENHERTU的治疗。
减少剂量后,请勿重新增加ENHERTU剂量。
如果延迟或错过了计划的剂量,请尽快给药;不要等到下一个计划周期。调整给药时间表以维持两次给药之间的3周间隔。以患者在最近一次输液中耐受的剂量和速率进行输液。
表1:减量时间表
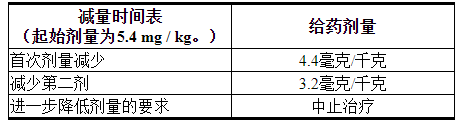
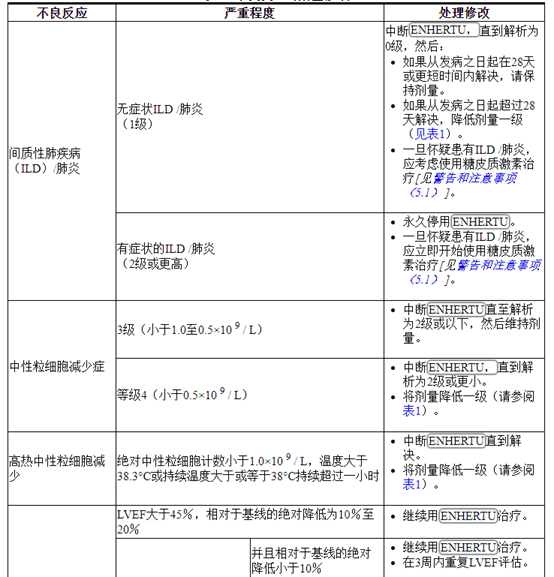
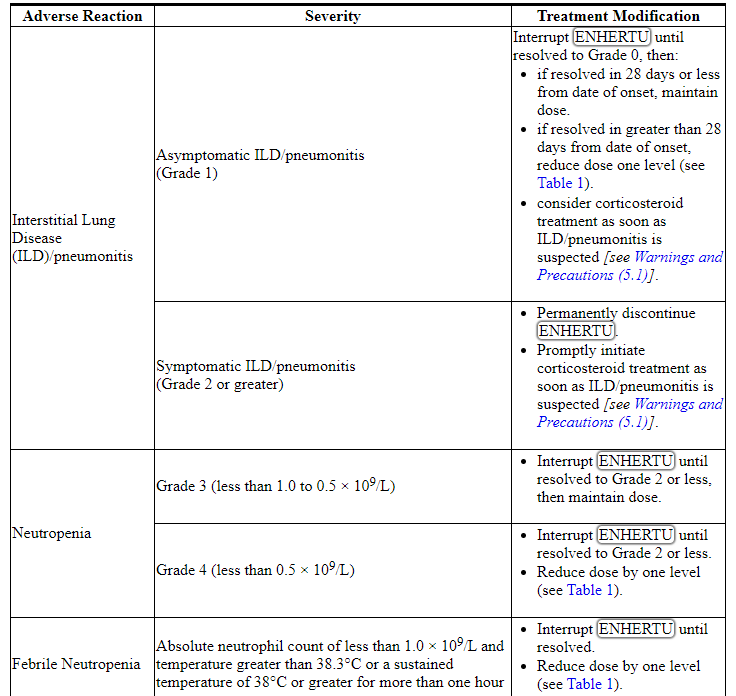
表2:不良反应的剂量修改
2.3准备管理
为了防止用药错误,请检查小瓶标签,以确保正在准备和使用的药物为ENHERTU(fam-曲妥珠单抗deruxtecan-nxki),而不是曲妥珠单抗或ado-曲妥珠单抗emtansine。
静脉输注前,重新配制并进一步稀释ENHERTU。使用适当的无菌技术。
ENHERTU(fam-trastuzumab deruxtecan-nxki)是一种细胞毒性药物。请遵循适用的特殊处理和处置程序。1个
重组
- 稀释前立即复溶。
- 全剂量可能需要一个以上的小瓶。计算剂量(毫克),重构的总体积ENHERTU的解决方案所需的,和小瓶(一个或多个)的数量ENHERTU需要[见剂量和给药方法(2.2) ]。
- 通过使用无菌注射器将5 mL无菌注射用水USP缓慢注入每个小瓶中,重新配制每个100 mg的小瓶,以达到20 mg / mL的最终浓度。
- 轻轻旋转小瓶,直到完全溶解。不要摇晃。
- 只要溶液和容器允许,在给药前应目视检查肠胃外药品中是否有颗粒物和变色。溶液应透明无色至浅黄色。如果观察到可见的颗粒或溶液混浊或变色,请勿使用。
- 如果不立即使用,请在复原后从避光处将重构的ENHERTU小瓶在2°C至8°C(36°F至46°F)的冰箱中存放长达24小时。不要冻结。
- 该产品不含防腐剂。冷藏24小时后,丢弃未使用的ENHERTU。
稀释
- 在装有100 mL 5%葡萄糖注射液USP的静脉输液袋中稀释计算得出的ENHERTU体积。请勿使用USP氯化钠注射液。ENHERTU与由聚氯乙烯或聚烯烃(乙烯和聚丙烯的共聚物)制成的输液袋兼容。
- 轻轻颠倒输液袋以彻底混合溶液。不要摇晃。
- 盖上输液袋以避光。
- 如果不立即使用,则在室温下最多可存放4个小时(包括准备和输注),或在2°C至8°C(36°F至46°F)的冰箱中保存24小时,并避免光照。不要冻结。
- 丢弃小瓶中所有未使用的部分。
行政
- 如果将制备的输注溶液冷藏保存(2°C至8°C [36°F至46°F]),则在给药前应使溶液达到室温。
- 辖ENHERTU静脉输液仅由聚烯烃或聚丁二烯的输液装置和0.20或0.22微米在线聚醚砜(PES)或聚砜(PS)滤波器。请勿静脉推注或推注。
- 请勿将ENHERTU与其他药物混合或通过同一静脉注射线给药其他药物。
3剂型和强度
注射用:100毫克fam-trastuzumab deruxtecan-nxki,白色至淡黄色白色冻干粉末,装在单剂量小瓶中,用于复溶和进一步稀释
4禁忌症
无。
5警告和注意事项
5.1间质性肺疾病/肺炎
用ENHERTU 治疗的患者可能会发生严重,威胁生命或致命的间质性肺病(ILD),包括肺炎[见不良反应(6.1) ]。在临床研究中,在用ENHERTU治疗的234例不可切除或转移性HER2阳性乳腺癌患者中,有9%的患者发生了ILD。在接受ENHERTU治疗的患者中,发生ILD和/或肺炎导致的致命结果发生率为2.6%。首次发病的中位时间为4.1个月(范围:1.2至8.3)。
劝告患者立即报告咳嗽,呼吸困难,发烧和/或任何新的或恶化的呼吸道症状。监视患者ILD的体征和症状。及时调查ILD的证据。通过影像学检查评估可疑ILD患者。考虑咨询肺科医生。对于无症状(1级)ILD,应考虑使用皮质类固醇治疗(例如≥0.5mg / kg泼尼松龙或同等剂量)。扣压ENHERTU直至痊愈[见剂量和给药方法(2.2) ]。如果出现症状性ILD(2级或更高),应立即开始皮质类固醇激素治疗(例如≥1 mg / kg泼尼松龙或同等剂量)。改善后,逐渐减少(例如4周)。永久停用ENHERTU诊断为有症状(2级或更高)ILD的患者[见剂量和用法(2.2) ]。
5.2中性粒细胞减少
使用ENHERTU治疗的患者可能会发生严重的中性粒细胞减少症,包括高热性中性粒细胞减少症。在接受ENHERTU治疗的234例无法切除或转移性HER2阳性乳腺癌患者中,据报道有30%的患者中性粒细胞计数减少,而16%的患者发生3级或4级事件。首次发作的中位时间为1.4个月(范围:0.3至18.2)。据报道1.7%的患者出现发热性中性粒细胞减少。
在开始ENHERTU之前和每次给药之前,并按照临床指示监测全血细胞计数。根据中性粒细胞减少症的严重程度,ENHERTU可能需要中断或降低剂量[见剂量和用法(2.2) ]。
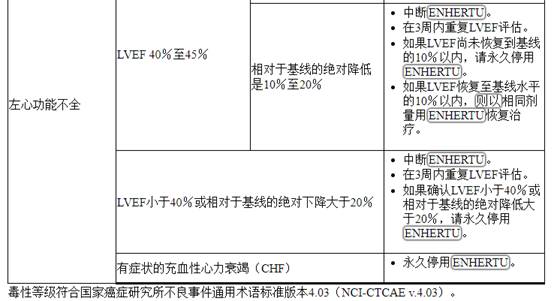
5.3左心室功能不全
接受ENHERTU治疗的患者发生左心功能不全的风险可能增加。包括ENHERTU在内的抗HER2治疗已观察到左室射血分数(LVEF)降低。在接受ENHERTU治疗的234例不可切除或转移性HER2阳性乳腺癌患者中,有2例(0.9%)无症状LVEF降低。对于在开始治疗前有临床显着性心脏病或LVEF少于50%的病史的患者,尚未进行ENHERTU治疗的研究。
在开始ENHERTU之前和临床治疗期间应定期评估LVEF 。通过治疗中断来控制LVEF降低。如果证实 LVEF小于40%或从基线绝对下降大于20%,则永久终止ENHERTU。症状性充血性心力衰竭(CHF)患者永久停用ENHERTU [请参阅剂量和给药方法(2.2) ]。
5.4胚胎-胎儿毒性
根据其作用机理,ENHERTU对孕妇服用可引起胎儿伤害。在上市后的报告中,怀孕期间使用HER2定向抗体导致羊水过少的病例,表现为致命的肺发育不全,骨骼异常和新生儿死亡。根据其作用的机制,拓扑异构酶抑制剂组分ENHERTU,DXD,也会造成当给予孕妇,因为它是基因毒性和目标活跃分裂细胞胚胎-胎儿危害[见特殊人群中使用(8.1) ,临床药理学(12.1),非临床毒理学(13.1) ]。告知患者胎儿的潜在危险。
在开始ENHERTU之前验证具有生殖潜力的女性的怀孕状况。建议有生殖潜力的女性在治疗期间和最后一次服用ENHERTU后 7个月内使用有效避孕。建议男性患者繁殖潜力与治疗过程中使用的有效避孕的女性伴侣ENHERTU和的最后一次给药后至少4个月ENHERTU [见特殊人群中使用(8.1,8.3)]。
6不良反应
标签上其他地方描述了以下临床上显着的不良反应:
- 间质性肺疾病/肺炎[请参阅警告和注意事项(5.1) ]
- 中性粒细胞减少症[请参阅警告和注意事项(5.2) ]
- 左心室功能障碍[请参阅警告和注意事项(5.3) ]
6.1临床试验经验
由于临床试验是在变化很大的条件下进行的,因此不能将在某种药物的临床试验中观察到的不良反应率直接与另一种药物的临床试验中观察到的不良反应率进行比较,并且可能无法反映实际中观察到的不良反应率。
通过对234例不可切除或转移性HER2阳性乳腺癌患者进行汇总分析,评估了ENHERTU的安全性,这些患者在DESTINY-Breast01和研究DS8201-A-J101(NCT02564900)中接受了至少一剂ENHERTU 5.4 mg / kg。ENHERTU每三周一次静脉输注。中位治疗时间为7个月(范围:0.7至31)。
在234名患者中,中位年龄为56岁(范围:28-96岁),其中74%的患者<65岁,女性的患者为99.6%,大多数为白人(51%)或亚裔(42%) )。患者在基线时的ECOG表现状态为0(58%)或1(42%)。94%患有内脏疾病,31%患有骨转移,13%患有脑转移。
接受ENHERTU治疗的患者中有20%发生严重的不良反应。接受ENHERTU的患者中> 1%的严重不良反应为间质性肺疾病,肺炎,呕吐,恶心,蜂窝组织炎,低血钾和肠梗阻。由不良反应引起的死亡发生在4.3%的患者中,包括间质性肺疾病(2.6%),并且每位患者(0.4%)发生以下事件:急性肝衰竭/急性肾损伤,一般身体健康恶化,肺炎和失血性休克。
9%的患者永久停用ENHERTU,其中ILD占6%。ENHERTU治疗的患者中有33%因不良反应导致剂量中断。与剂量中断相关的最常见不良反应(> 2%)是中性粒细胞减少,贫血,血小板减少,白细胞减少,上呼吸道感染,疲劳,恶心和ILD。用ENHERTU治疗的患者中有18%的患者出现剂量减少。与剂量减少相关的最常见不良反应(> 2%)是疲劳,恶心和中性粒细胞减少。
最常见的不良反应(频率≥20%)为恶心,疲劳,呕吐,脱发,便秘,食欲下降,贫血,中性粒细胞减少,腹泻,白细胞减少,咳嗽和血小板减少。
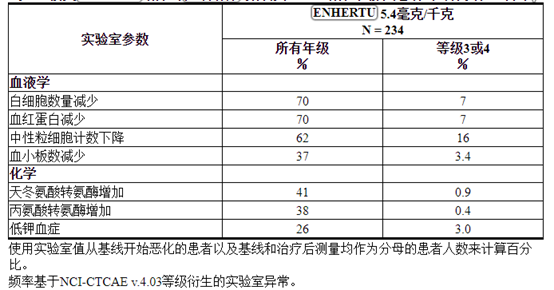
表3和表4总结了在ENHERTU治疗的患者中观察到的常见不良反应和实验室异常。
表3:DESTINY-Breast01和研究DS8201-A-J101中患者的常见不良反应(≥10%所有等级或≥2%3或4级)
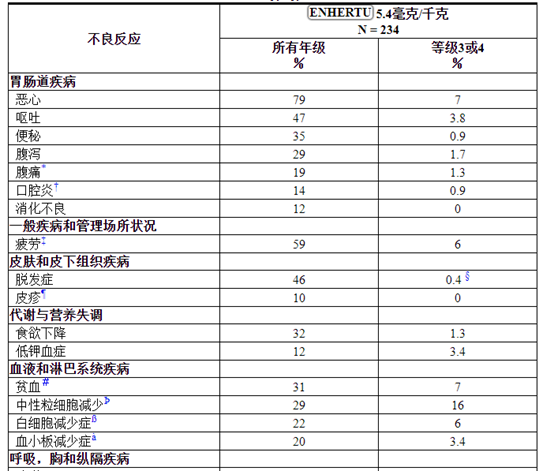

少于10%的患者报告的其他与临床相关的不良反应是:
- 损伤,中毒和程序并发症:与输液有关的反应(2.6%)
- 血液和淋巴系统疾病:发热性中性粒细胞减少症(1.7%)
表4:接受ENHERTU治疗的无法切除或转移性HER2阳性乳腺癌患者的部分实验室异常
6.2免疫原性
与所有治疗性蛋白质一样,具有免疫原性的潜力。抗体形成的检测高度依赖于测定的灵敏度和特异性。另外,在测定中观察到的抗体(包括中和抗体)阳性的发生率可能受到多种因素的影响,包括测定方法,样品处理,样品收集的时间,伴随用药和基础疾病。由于这些原因,将下述研究中的ENHERTU抗体发生率与其他研究或其他产品中抗体发生率进行比较可能会产生误导。
在所有剂量下均接受ENHERTU治疗的0.6%(4/640)患者中出现了治疗诱导的抗家族曲妥珠单抗deruxtecan-nxki抗体(ADA)。由于对ADA呈阳性的患者数量有限,因此无法得出有关免疫原性对功效或安全性的潜在影响的结论。另外,尚未评估抗ENHERTU抗体的中和活性。
8在特定人群中的使用
8.1怀孕
风险摘要
根据其作用机理,ENHERTU对孕妇服用可引起胎儿伤害。没有关于孕妇使用ENHERTU的可用数据。在上市后的报告中,妊娠期间使用HER2定向抗体导致羊水过少的病例表现为致命的肺发育不全,骨骼异常和新生儿死亡[参见数据 ]。根据其作用的机制,拓扑异构酶抑制剂组分ENHERTU,DXD,也会造成当给予孕妇,因为它是基因毒性和目标活跃分裂细胞胚胎-胎儿危害[见临床药理学(12.1) ,非临床毒理学(13.1 )]。告知患者胎儿的潜在危险。
如果在孕妇中使用ENHERTU,或者患者在最后一次使用ENHERTU 后7个月内怀孕,则有临床考虑 [请参见临床注意事项 ]。
对于指定人群,主要出生缺陷和流产的估计背景风险尚不清楚。所有怀孕都有出生缺陷,流产或其他不良后果的背景风险。在美国普通人群中,在临床上公认的怀孕中,主要先天缺陷和流产的估计背景风险分别为2-4%和15-20%。
临床注意事项
胎儿/新生儿不良反应
监测怀孕期间或受孕前7个月内因羊水过少而接受ENHERTU治疗的妇女。如果发生羊水过少,请进行适合胎龄并符合社区护理标准的胎儿测试。
数据
人数据
没有关于孕妇使用ENHERTU的可用数据。在孕妇接受HER2定向抗体的上市后报告中,曾报道羊水过少的病例,这些病例表现为致命的肺发育不全,骨骼异常和新生儿死亡。这些病例报告描述了孕妇中的羊水过少,该孕妇单独或与化学疗法联合接受HER2定向抗体。在某些情况下,在停止使用HER2定向抗体后羊水指数增加。
动物资料
没有使用fam-trastuzumab deruxtecan-nxki进行的动物生殖或发育毒性研究。
8.2哺乳
风险摘要
没有关于母乳中是否存在fam-trastuzumab deruxtecan-nxki,对母乳喂养的孩子的影响或对牛奶产量的影响的数据。由于母乳喂养的孩子可能会出现严重的不良反应,因此建议女性在进行ENHERTU治疗期间以及末次用药后7个月内不要母乳喂养。
8.3生殖潜力的雌雄
验孕
在开始ENHERTU之前确认具有生殖潜能的女性的怀孕状况。
避孕
女性
给孕妇服用时,ENHERTU可能会造成胎儿伤害[请参见“ 在特定人群中使用(8.1)” ]。劝告有生殖潜力的女性在使用ENHERTU治疗期间以及末次服药后至少7个月内使用有效的避孕方法。
雄性
由于有潜在的遗传毒性,建议有生殖潜能的女性伴侣的男性患者在接受ENHERTU治疗期间以及最后一次给药后至少4个月内使用有效的避孕方法[请参阅非临床毒理学(13.1) ]。
不孕症
根据动物毒性研究的结果,ENHERTU可能会损害男性的生殖功能和生育能力[请参见非临床毒理学(13.1) ]。
8.4小儿使用
在儿科患者中尚未确定ENHERTU的安全性和有效性。
8.5老年用途
在用ENHERTU 5.4 mg / kg 治疗的234例HER2阳性乳腺癌患者中,有26%为65岁或以上,而5%为75岁或以上。与年轻患者相比,≥65岁的患者之间未观察到总体疗效差异。与年龄较小的患者(42%)相比,年龄在65岁或以上的患者(53%)观察到的3-4级不良反应发生率更高。
8.6肾功能不全
对于轻度(肌酐清除率(CLcr)≥60和<90 mL / min)或中度(CLcr≥30和<60 mL / min)肾功能不全的患者,无需调整ENHERTU的剂量[参见临床药理学(12.3) ]。没有严重肾功能不全患者的数据。
8.7肝功能不全
轻度(总胆红素≤ULN并且任何AST> ULN或总胆红素> 1至1.5倍ULN和任何AST)或中度(总胆红素> 1.5至3倍ULN和任何AST)的患者无需调整ENHERTU剂量损害。在中度肝功能不全的患者中,由于潜在增加的暴露,应密切监测与拓扑异构酶抑制剂DXd相关的毒性的增加[参见剂量和给药方法(2.2) ]。严重(总胆红素> ULN的3到10倍,且任何AST)肝功能不全的患者无可用数据[见临床药理学(12.3) ]。
11说明
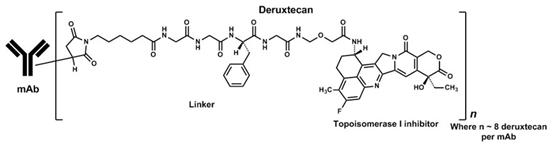
Fam-trastuzumab deruxtecan-nxki是HER2定向抗体和拓扑异构酶抑制剂的结合物。Fam-trastuzumab deruxtecan-nxki是一种抗体-药物偶联物(ADC),由三个部分组成:1)与2)拓扑异构酶抑制剂共价连接的人源化抗HER2 IgG1单克隆抗体(mAb),通过3)基于四肽的可裂解的接头。Deruxtecan由蛋白酶可裂解的马来酰亚胺四肽接头和拓扑异构酶抑制剂DXd组成,后者是exatecan衍生物。
该抗体是通过重组DNA技术在中国仓鼠卵巢细胞中产生的,拓扑异构酶抑制剂和接头是通过化学合成产生的。大约8个分子的deruxtecan分子附着在每个抗体分子上。Fam-曲妥珠单抗deruxtecan-nxki具有以下结构:
ENHERTU(fam-trastuzumab deruxtecan-nxki)是单剂量小瓶中无菌,白色至淡黄色无防腐剂的冻干粉末。每个小瓶可递送100 mg的fam-曲妥珠单抗deruxtecan-nxki,L-组氨酸(4.45 mg),L-组氨酸盐酸盐一水合物(20.2 mg),聚山梨酸酯80(1.5 mg)和蔗糖(450 mg)。用5 mL无菌注射用水(USP)复溶后,fam-曲妥珠单抗deruxtecan-nxki的最终浓度为20 mg / mL,pH值为5.5。稀释后通过静脉输注给予所得溶液。
12临床药理学
12.1行动机制
Fam-曲妥珠单抗deruxtecan-nxki是HER2定向的抗体-药物偶联物。该抗体是人源化抗HER2 IgG1。小分子DXd是拓扑异构酶I抑制剂,通过可裂解的接头与抗体连接。与肿瘤细胞上的HER2结合后,fam-trastuzumab deruxtecan-nxki通过溶酶体酶进行内在化和细胞内接头裂解。释放后,可透膜的DXd引起DNA损伤和凋亡细胞死亡。
12.2药效学
心脏电生理学
在开放标签的单臂研究中,多次服用ENHERTU(每3周6.4 mg / kg,是推荐剂量的1.2倍)并未对QTc间隔表现出较大的平均影响(即> 20 ms)。 51例HER2表达转移性乳腺癌患者。
12.3药代动力学
评估了fam-trastuzumab deruxtecan-nxki在癌症患者中的药代动力学。单次给药后,fam-曲妥珠单抗deruxtecan-nxki和释放的拓扑异构酶抑制剂(DXd)的暴露量(C max和AUC)在3.2 mg / kg至8 mg / kg的剂量范围内成比例增加(约为推荐剂量的0.6至1.5倍)剂量)。在推荐剂量的ENHERTU下,几何平均值(变异系数[CV]%)C maxfam-曲妥珠单抗deruxtecan-nxki和DXd的AUC分别为122μg/ mL(20%)和4.4 ng / mL(40%),并且fam-曲妥珠单抗deruxtecan-nxki和DXd的AUC为735μg∙day / mL(根据人群药代动力学分析,分别为31%)和28 ng∙day / mL(38%)。稳定状态下(循环3),fam-trastuzumab deruxtecan-nxki的积累约为35%。
分配
根据人群药代动力学分析,fam-曲妥珠单抗deruxtecan-nxki 的中央区室(V c)的估计分布量为2.77L。
对于人类而言,体外DXd血浆蛋白结合率约为97%,血液与血浆的比率约为0.6。
消除
fam-曲妥珠单抗deruxtecan-nxki 的中值消除半衰期(t 1/2)约为5.7天。根据人群药代动力学分析,fam-曲妥珠单抗deruxtecan-nxki的全身清除率估计为0.42 L /天。
DXd 的中值表观消除半衰期(t 1/2)约为5.8天。根据人群药代动力学分析,DXd的表观全身清除率估计为19.2 L / h。
代谢
预期人源化HER2 IgG1单克隆抗体将以与内源IgG相同的方式通过分解代谢途径降解为小肽和氨基酸。
在体外,DXd主要由CYP3A4代谢。
特定人群
对于年龄(23-96岁),种族(亚洲[n = 291]和非亚洲[n = 221]),性别,体重,fam-trastuzumab deruxtecan-nxki或DXd的药代动力学没有临床显着差异。 (34.6-125.4 kg),轻度(总胆红素≤ULN,任何AST> ULN或总胆红素> 1至1.5倍ULN,任何AST,n = 215),肝功能不全,轻度(肌酐清除率[CLcr]≥60和<90 mL / min,n = 206)或中度(CLcr≥30和<60 mL / min; n = 58)肾功能不全,基于人群药代动力学分析。
尚不清楚fam-trastuzumab deruxtecan-nxki或DXd在中度至重度肝功能不全(任何AST中总胆红素> 1.5 ULN)或严重肾功能不全(CLcr <30 mL / min)的患者中的药代动力学。
药物相互作用研究
临床研究
CYP3A抑制剂对DXd的影响:联合使用伊曲康唑,一种强效CYP3A抑制剂和多剂量的ENHERTU,可使fam-trastuzumab deruxtecan-nxki的稳态AUC 0-17天增加11%,而DXd则增加18%。这些变化的影响在临床上没有意义。
OATP抑制剂对DXd的影响:利托那韦(OATP1B / CYP3A的双重抑制剂)与多次剂量的ENHERTU并用会增加fam-trastuzumab deruxtecan-nxki的稳态AUC 0-17天和 DXd的22%。这些变化的影响在临床上没有意义。
体外研究
DXd对CYP酶的影响: DXd不抑制CYP1A2,CYP2B6,CYP2C8,CYP2C9,CYP2C19,CYP2D6和CYP3A,也不会诱导CYP1A2,CYP2B6或CYP3A。
DXd对转运蛋白的影响:在临床相关浓度下(稳态C max为〜0.2 µmol / L),DXd抑制OAT1(IC 50值为12.7 µmol / L),OAT3,OCT1,OCT2,OATP1B1的可能性很低。 (IC 50值为14.4 µmol / L),OATP1B3,MATE1,MATE2-K,P-gp,BCRP或BSEP转运蛋白。
其他药物对DXd的影响: DXd是OATP1B1,OATP1B3,MATE2-K,P-gp,MRP1和BCRP的底物。
13毒理学
13.1致癌,诱变,生育力受损
尚未对fam-trastuzumab deruxtecan-nxki进行致癌性研究。
fam-trastuzumab deruxtecan-nxki(DXd)的拓扑异构酶抑制剂成分在体内大鼠骨髓微核试验和体外中国仓鼠肺染色体畸变试验中均具有致死性,而在体外细菌反向突变试验中则无致突变性。
尚未对fam-trastuzumab deruxtecan-nxki进行生育力研究。在对大鼠进行的为期六周的重复剂量毒性研究中,fam-trastuzumab deruxtecan-nxki的静脉内给药导致精子保留率分别为20 mg / kg和60 mg / kg(约为人类建议剂量5.4 mg / kg的4和9倍) kg分别基于AUC)。以197 mg / kg的剂量观察到睾丸和附睾重量减少,睾丸的肾小管萎缩/变性以及附睾中的精子数量减少(基于AUC的人推荐剂量5.4 mg / kg的19倍)。在对猴子进行的为期三个月的重复剂量毒性研究中,fam-trastuzumab deruxtecan-nxki的静脉内给药导致≥30 mg / kg的生精小管阶段V至VI的睾丸中圆形精子数量减少。人类推荐剂量为5。4 mg / kg(基于AUC)。在三个月的恢复期结束时,已在猴子中观察到可逆性的证据。
14临床研究
14.1转移性乳腺癌
在一项多中心单臂试验DESTINY-Breast01(NCT03248492)研究中评估了ENHERTU的疗效,该试验招募了184名患有HER2阳性,不可切除和/或转移性乳腺癌的女性患者,这些患者曾接受过两次或更多次抗HER2的治疗。疗法。筛选时排除患者有治疗ILD史或当前ILD史。患者也因临床上明显的心脏病史,活跃的脑转移病史和ECOG表现状态> 1而被排除在外。HER2表达是基于入组前在中央实验室测试的档案组织而定义的HER2阳性定义为HER2 IHC 3+或ISH阳性。
患者每三周静脉输注ENHERTU 5.4 mg / kg,直到出现不可接受的毒性或疾病进展。每6周进行一次肿瘤成像,基线时脑转移的患者必须进行脑部CT / MRI检查。主要疗效结果通过使用RECIST v1.1的独立中央评估评估的客观缓解率(ORR)和反应持续时间(DOR)。
中位年龄为55岁(范围:28-96);76%的患者<65岁。184例患者均为女性,大多数为白人(55%)或亚裔(38%)。患者在基线时的ECOG表现状态为0(55%)或1(44%)。92%患有内脏疾病,29%患有骨转移,13%患有脑转移。53%的激素受体阳性。目标病变的直径总和在42%的情况下<5 cm,在50%的情况下≥5 cm(8%的患者无法通过中央检查评估)。
在局部晚期/转移环境中,先前癌症治疗方案的中位数为5(范围:2-17)。
所有患者均接受过曲妥珠单抗,阿多-曲妥珠单抗坦坦碱治疗,其中66%的患者曾接受过帕妥珠单抗治疗。
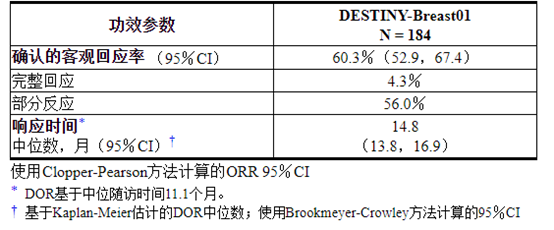
疗效结果总结在表5中。
表5:DESTINY-Breast01中独立中央评价的疗效结果
15参考
1.OSHA危险药物。OSHA。http://www.osha.gov/SLTC/hazardousdrugs/index.html
16供应/存储和处理方式
16.1供应/存储方式
注射用ENHERTU(fam-trastuzumab deruxtecan-nxki)为白色至淡黄色白色冻干粉末,供应:
将小瓶放在原始纸箱中的2°C至8°C(36°F至46°F)的冰箱中,以防光照,直到重新配制为止。不要冻结。请勿摇动重构或稀释的溶液[参见剂量和用法(2.3) ]。
16.2特殊处理
ENHERTU(fam-trastuzumab deruxtecan-nxki)是一种细胞毒性药物。请遵循适用的特殊处理和处置程序。1个
17患者咨询信息
建议患者阅读FDA批准的患者标签(用药指南)。
间质性肺疾病
- 告知患者严重或致命的ILD风险。建议患者就以下任何情况立即联系其医疗保健提供者:咳嗽,气短,发烧或其他新的或恶化的呼吸道症状[请参阅警告和注意事项(5.1) ]。
中性粒细胞减少症
- 告知患者出现中性粒细胞减少症的可能性,并在发烧时立即联系其医疗服务提供者,尤其是与任何感染迹象有关的情况[见警告和注意事项(5.2) ]。
左心功能不全
- 建议患者立即就以下任何情况与医疗保健提供者联系:新发或呼吸急促,咳嗽,疲劳,脚踝/腿肿胀,心pal,体重突然增加,头昏眼花,失去知觉[请参阅警告和注意事项(5.3 ) ]。
胚胎-胎儿毒性
- 告知女性患者胎儿的潜在危险。劝告女性患者就已知或疑似怀孕联系其医护人员[请参阅警告和注意事项(5.4),在特定人群中使用(8.1) ]。
- 劝告有生殖潜力的女性在ENHERTU治疗期间以及最后一次给药后至少7个月内使用有效的避孕方法(请参见“ 在特定人群中使用(8.3) ”)。
- 建议具有生殖潜力的女性伴侣的男性患者在进行ENHERTU治疗期间以及最后一次给药后至少4个月内使用有效的避孕药[请参见在特定人群中使用(8.3) ]。
哺乳期
- 劝告妇女在治疗期间和末次使用 ENHERTU 后7个月内不要进行母乳喂养[见在特定人群中使用(8.2) ]。
不孕症
- 向有生殖能力的男性建议,ENHERTU可能会损害生育能力[请参阅在特定人群中使用(8.3) ]。
制造商:日本
第一三共公司(Basking Ridge),新泽西州07920美国许可证号2128
市场销售商:
Daiichi Sankyo,Inc.(位于Basking Ridge,NJ 07920)和AstraZeneca Pharmaceuticals LP,Wilmington,DE 19850ENHERTU ®是第一三共制药股份有限公司的注册商标。
©2019第一三共株式会社USPI-ENH-C1-1219-r001


主要显示屏-100毫克瓶装纸箱
仅NDC 65597-406-01 RxENHERTU ®
(FAM-曲妥珠单抗deruxtecan-nxki)注射用
每瓶100毫克
对于静脉输注,仅
向每位患者分配随附的用药指南。
给药前复溶并稀释
单剂量小瓶
丢弃未使用的部分注意:细胞毒剂
保持冷藏
1瓶
第一三共
阿斯利康
【备注】以上内容仅供参考,不作为用药依据,详情请参照药品附带说明书。
-
本说明书来源于:美国FDA网站
温馨提醒:
①建议您用 谷歌浏览器 在电脑上或手机 打开以上链接,就可以自动翻译成简体中文,而且翻译的还比较准确。
②本说明书仅供参考,最新的说明书详见药品附带的说明书。
WARNING: INTERSTITIAL LUNG DISEASE and EMBRYO-FETAL TOXICITY
- Interstitial Lung Disease (ILD) and pneumonitis, including fatal cases, have been reported with ENHERTU. Monitor for and promptly investigate signs and symptoms including cough, dyspnea, fever, and other new or worsening respiratory symptoms. Permanently discontinue ENHERTU in all patients with Grade 2 or higher ILD/pneumonitis. Advise patients of the risk and the need to immediately report symptoms [see Dosage and Administration (2.2), Warnings and Precautions (5.1)].
- Embryo-Fetal Toxicity: Exposure to ENHERTU during pregnancy can cause embryo-fetal harm. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.4), Use in Specific Populations (8.1, 8.3)].
1 INDICATIONS AND USAGE
ENHERTU is indicated for the treatment of adult patients with unresectable or metastatic HER2-positive breast cancer who have received two or more prior anti-HER2-based regimens in the metastatic setting.
This indication is approved under accelerated approval based on tumor response rate and duration of response [see Clinical Studies (14.1)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage and Schedules
Do not substitute ENHERTU for or with trastuzumab or ado-trastuzumab emtansine.
The recommended dosage of ENHERTU is 5.4 mg/kg given as an intravenous infusion once every 3 weeks (21-day cycle) until disease progression or unacceptable toxicity.
First infusion: Administer infusion over 90 minutes.
Subsequent infusions: Administer over 30 minutes if prior infusions were well tolerated.
Slow or interrupt the infusion rate if the patient develops infusion-related symptoms.
Permanently discontinue ENHERTU in case of severe infusion reactions.
2.2 Dose Modifications
Management of adverse reactions may require temporary interruption, dose reduction, or treatment discontinuation of ENHERTU as described in Tables 1 and 2.
Do not re-escalate the ENHERTU dose after a dose reduction is made.
If a planned dose is delayed or missed, administer as soon as possible; do not wait until the next planned cycle. Adjust the schedule of administration to maintain a 3-week interval between doses. Administer the infusion at the dose and rate the patient tolerated in the most recent infusion.
Table 1: Dose Reduction Schedule
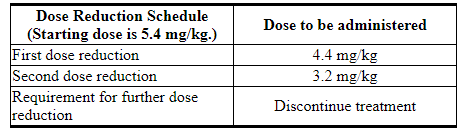
Table 2: Dose Modifications for Adverse Reactions
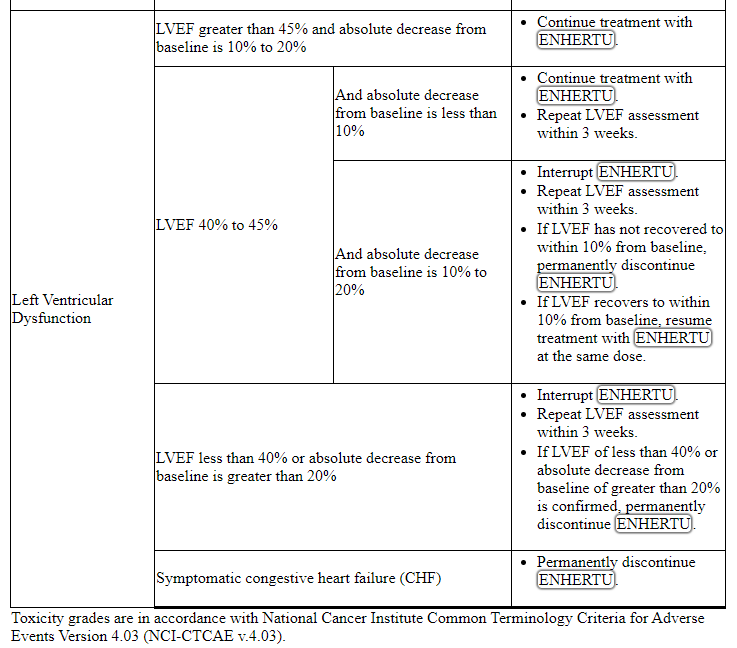
2.3 Preparation for Administration
In order to prevent medication errors, check the vial labels to ensure that the drug being prepared and administered is ENHERTU (fam-trastuzumab deruxtecan-nxki) and not trastuzumab or ado-trastuzumab emtansine.
Reconstitute and further dilute ENHERTU prior to intravenous infusion. Use appropriate aseptic technique.
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a cytotoxic drug. Follow applicable special handling and disposal procedures.1
Reconstitution
- Reconstitute immediately before dilution.
- More than one vial may be needed for a full dose. Calculate the dose (mg), the total volume of reconstituted ENHERTU solution required, and the number of vial(s) of ENHERTU needed [see Dosage and Administration (2.2)].
- Reconstitute each 100 mg vial by using a sterile syringe to slowly inject 5 mL of Sterile Water for Injection, USP into each vial to obtain a final concentration of 20 mg/mL.
- Swirl the vial gently until completely dissolved. Do not shake.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. The solution should be clear and colorless to light yellow. Do not use if visible particles are observed or if the solution is cloudy or discolored.
- If not used immediately, store the reconstituted ENHERTU vials in a refrigerator at 2°C to 8°C (36°F to 46°F) for up to 24 hours from the time of reconstitution, protected from light. Do not freeze.
- The product does not contain a preservative. Discard unused ENHERTU after 24 hours refrigerated.
Dilution
- Dilute the calculated volume of reconstituted ENHERTU in an intravenous infusion bag containing 100 mL of 5% Dextrose Injection, USP. Do not use Sodium Chloride Injection, USP. ENHERTU is compatible with an infusion bag made of polyvinylchloride or polyolefin (copolymer of ethylene and polypropylene).
- Gently invert the infusion bag to thoroughly mix the solution. Do not shake.
- Cover the infusion bag to protect from light.
- If not used immediately, store at room temperature for up to 4 hours including preparation and infusion, or in a refrigerator at 2°C to 8°C (36°F to 46°F) for up to 24 hours, protected from light. Do not freeze.
- Discard any unused portion left in the vial.
Administration
- If the prepared infusion solution was stored refrigerated (2°C to 8°C [36°F to 46°F]), allow the solution to reach room temperature prior to administration.
- Administer ENHERTU as an intravenous infusion only with an infusion set made of polyolefin or polybutadiene and a 0.20 or 0.22 micron in-line polyethersulfone (PES) or polysulfone (PS) filter. Do not administer as an intravenous push or bolus.
- Do not mix ENHERTU with other drugs or administer other drugs through the same intravenous line.
3 DOSAGE FORMS AND STRENGTHS
For injection: 100 mg of fam-trastuzumab deruxtecan-nxki as a white to yellowish white lyophilized powder in a single-dose vial for reconstitution and further dilution
4 CONTRAINDICATIONS
None.
5 WARNINGS AND PRECAUTIONS
5.1 Interstitial Lung Disease/Pneumonitis
Severe, life-threatening, or fatal interstitial lung disease (ILD), including pneumonitis, can occur in patients treated with ENHERTU [see Adverse Reactions (6.1)]. In clinical studies, of the 234 patients with unresectable or metastatic HER2-positive breast cancer treated with ENHERTU, ILD occurred in 9% of patients. Fatal outcomes due to ILD and/or pneumonitis occurred in 2.6% of patients treated with ENHERTU. Median time to first onset was 4.1 months (range: 1.2 to 8.3).
Advise patients to immediately report cough, dyspnea, fever, and/or any new or worsening respiratory symptoms. Monitor patients for signs and symptoms of ILD. Promptly investigate evidence of ILD. Evaluate patients with suspected ILD by radiographic imaging. Consider consultation with a pulmonologist. For asymptomatic (Grade 1) ILD, consider corticosteroid treatment (e.g., ≥0.5 mg/kg prednisolone or equivalent). Withhold ENHERTU until recovery [see Dosage and Administration (2.2)]. In cases of symptomatic ILD (Grade 2 or greater), promptly initiate corticosteroid treatment (e.g., ≥1 mg/kg prednisolone or equivalent). Upon improvement, follow by gradual taper (e.g., 4 weeks). Permanently discontinue ENHERTU in patients who are diagnosed with any symptomatic (Grade 2 or greater) ILD [see Dosage and Administration (2.2)].
5.2 Neutropenia
Severe neutropenia, including febrile neutropenia, can occur in patients treated with ENHERTU. Of the 234 patients with unresectable or metastatic HER2-positive breast cancer who received ENHERTU, a decrease in neutrophil count was reported in 30% of patients and 16% had Grade 3 or 4 events. Median time to first onset was 1.4 months (range: 0.3 to 18.2). Febrile neutropenia was reported in 1.7% of patients.
Monitor complete blood counts prior to initiation of ENHERTU and prior to each dose, and as clinically indicated. Based on the severity of neutropenia, ENHERTU may require dose interruption or reduction [see Dosage and Administration (2.2)].
5.3 Left Ventricular Dysfunction
Patients treated with ENHERTU may be at increased risk of developing left ventricular dysfunction. Left ventricular ejection fraction (LVEF) decrease has been observed with anti-HER2 therapies, including ENHERTU. In the 234 patients with unresectable or metastatic HER2-positive breast cancer who received ENHERTU, two cases (0.9%) of asymptomatic LVEF decrease were reported. Treatment with ENHERTU has not been studied in patients with a history of clinically significant cardiac disease or LVEF less than 50% prior to initiation of treatment.
Assess LVEF prior to initiation of ENHERTU and at regular intervals during treatment as clinically indicated. Manage LVEF decrease through treatment interruption. Permanently discontinue ENHERTU if LVEF of less than 40% or absolute decrease from baseline of greater than 20% is confirmed. Permanently discontinue ENHERTU in patients with symptomatic congestive heart failure (CHF) [see Dosage and Administration (2.2)].
5.4 Embryo-Fetal Toxicity
Based on its mechanism of action, ENHERTU can cause fetal harm when administered to a pregnant woman. In postmarketing reports, use of a HER2-directed antibody during pregnancy resulted in cases of oligohydramnios manifesting as fatal pulmonary hypoplasia, skeletal abnormalities, and neonatal death. Based on its mechanism of action, the topoisomerase inhibitor component of ENHERTU, DXd, can also cause embryo-fetal harm when administered to a pregnant woman because it is genotoxic and targets actively dividing cells [see Use in Specific Populations (8.1), Clinical Pharmacology (12.1), Nonclinical Toxicology (13.1)]. Advise patients of the potential risks to a fetus.
Verify the pregnancy status of females of reproductive potential prior to the initiation of ENHERTU. Advise females of reproductive potential to use effective contraception during treatment and for at least 7 months following the last dose of ENHERTU. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ENHERTU and for at least 4 months after the last dose of ENHERTU [see Use in Specific Populations (8.1, 8.3)].
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Interstitial Lung Disease / Pneumonitis [see Warnings and Precautions (5.1)]
- Neutropenia [see Warnings and Precautions (5.2)]
- Left Ventricular Dysfunction [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ENHERTU was evaluated in a pooled analysis of 234 patients with unresectable or metastatic HER2-positive breast cancer who received at least one dose of ENHERTU 5.4 mg/kg in DESTINY-Breast01 and Study DS8201-A-J101 (NCT02564900). ENHERTU was administered by intravenous infusion once every three weeks. The median duration of treatment was 7 months (range: 0.7 to 31).
In the pooled 234 patients, the median age was 56 years (range: 28-96), 74% of patients were <65 years, 99.6% of patients were female, and the majority were White (51%) or Asian (42%). Patients had an ECOG performance status of 0 (58%) or 1 (42%) at baseline. Ninety-four percent had visceral disease, 31% had bone metastases, and 13% had brain metastases.
Serious adverse reactions occurred in 20% of patients receiving ENHERTU. Serious adverse reactions in >1% of patients who received ENHERTU were interstitial lung disease, pneumonia, vomiting, nausea, cellulitis, hypokalemia, and intestinal obstruction. Fatalities due to adverse reactions occurred in 4.3% of patients including interstitial lung disease (2.6%), and the following events occurred in one patient each (0.4%): acute hepatic failure/acute kidney injury, general physical health deterioration, pneumonia, and hemorrhagic shock.
ENHERTU was permanently discontinued in 9% of patients, of which ILD accounted for 6%. Dose interruptions due to adverse reactions occurred in 33% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose interruption were neutropenia, anemia, thrombocytopenia, leukopenia, upper respiratory tract infection, fatigue, nausea, and ILD. Dose reductions occurred in 18% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose reduction were fatigue, nausea, and neutropenia.
The most common adverse reactions (frequency ≥20%) were nausea, fatigue, vomiting, alopecia, constipation, decreased appetite, anemia, neutropenia, diarrhea, leukopenia, cough, and thrombocytopenia.
Tables 3 and 4 summarize common adverse reactions and laboratory abnormalities observed in ENHERTU-treated patients.
Table 3: Common Adverse Reactions (≥10% All Grades or ≥2% Grades 3 or 4) in Patients in DESTINY-Breast01 and Study DS8201-A-J101
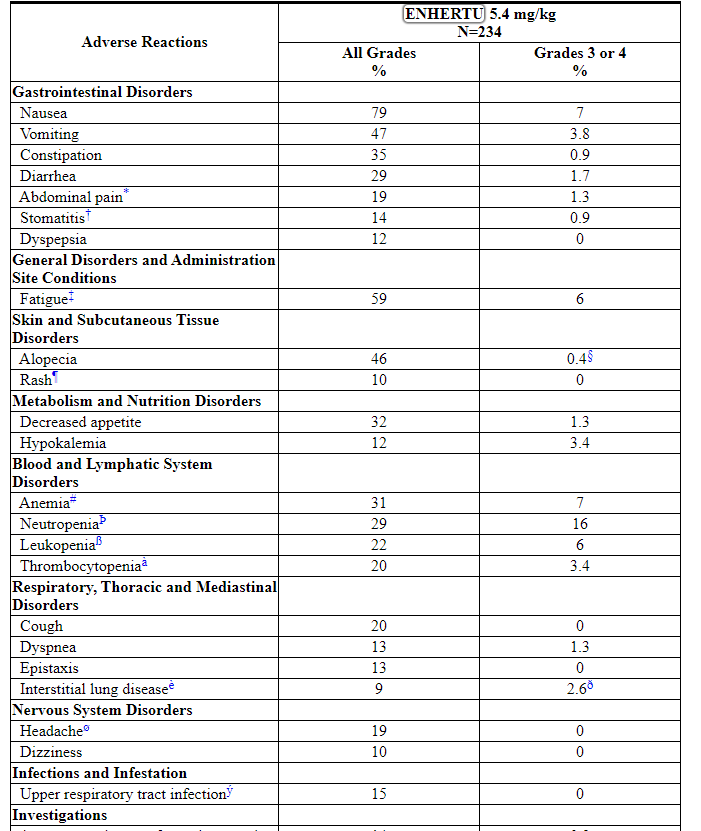
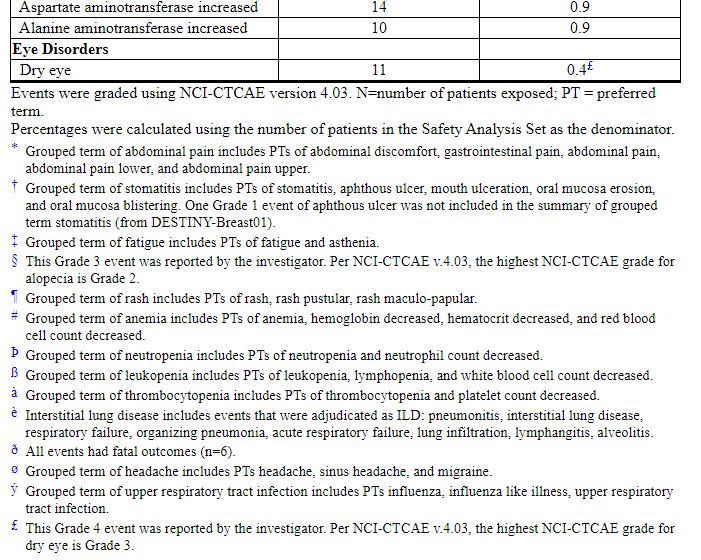
Other clinically relevant adverse reactions reported in less than 10% of patients were:
- Injury, Poisoning and Procedural Complications: infusion-related reactions (2.6%)
- Blood and Lymphatic System Disorders: febrile neutropenia (1.7%)
Table 4: Selected Laboratory Abnormalities in Patients with Unresectable or Metastatic HER2-positive Breast Cancer Treated with ENHERTU
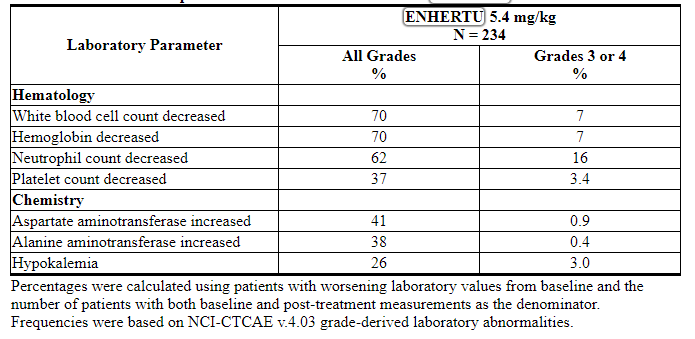
6.2 Immunogenicity
As with all therapeutic proteins, there is a potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparisons of the incidence of antibodies to ENHERTU in the studies described below with the incidence of antibodies in other studies or to other products may be misleading.
Treatment-induced anti-fam-trastuzumab deruxtecan-nxki antibodies (ADA) developed in 0.6% (4/640) patients who received ENHERTU across all doses. Due to the limited number of patients who tested positive for ADA, no conclusions can be drawn concerning a potential effect of immunogenicity on efficacy or safety. In addition, neutralizing activity of anti-ENHERTU antibodies has not been assessed.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on its mechanism of action, ENHERTU can cause fetal harm when administered to a pregnant woman. There are no available data on the use of ENHERTU in pregnant women. In postmarketing reports, use of a HER2-directed antibody during pregnancy resulted in cases of oligohydramnios manifesting as fatal pulmonary hypoplasia, skeletal abnormalities, and neonatal death [see Data]. Based on its mechanism of action, the topoisomerase inhibitor component of ENHERTU, DXd, can also cause embryo-fetal harm when administered to a pregnant woman because it is genotoxic and targets actively dividing cells [see Clinical Pharmacology (12.1), Nonclinical Toxicology (13.1)]. Advise patients of the potential risks to a fetus.
There are clinical considerations if ENHERTU is used in pregnant women, or if a patient becomes pregnant within 7 months following the last dose of ENHERTU [see Clinical Considerations].
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Monitor women who received ENHERTU during pregnancy or within 7 months prior to conception for oligohydramnios. If oligohydramnios occurs, perform fetal testing that is appropriate for gestational age and consistent with community standards of care.
Data
Human Data
There are no available data on the use of ENHERTU in pregnant women. In postmarketing reports in pregnant women receiving a HER2-directed antibody, cases of oligohydramnios manifesting as fatal pulmonary hypoplasia, skeletal abnormalities, and neonatal death have been reported. These case reports described oligohydramnios in pregnant women who received a HER2-directed antibody either alone or in combination with chemotherapy. In some case reports, amniotic fluid index increased after use of a HER2-directed antibody was stopped.
Animal Data
There were no animal reproductive or developmental toxicity studies conducted with fam-trastuzumab deruxtecan-nxki.
8.2 Lactation
Risk Summary
There is no data regarding the presence of fam-trastuzumab deruxtecan-nxki in human milk, the effects on the breastfed child, or the effects on milk production. Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during treatment with ENHERTU and for 7 months after the last dose.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Verify pregnancy status of females of reproductive potential prior to initiation of ENHERTU.
Contraception
Females
ENHERTU can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)]. Advise females of reproductive potential to use effective contraception during treatment with ENHERTU and for at least 7 months following the last dose.
Males
Because of the potential for genotoxicity, advise male patients with female partners of reproductive potential to use effective contraception during treatment with ENHERTU and for at least 4 months following the last dose [see Nonclinical Toxicology (13.1)].
Infertility
Based on findings in animal toxicity studies, ENHERTU may impair male reproductive function and fertility [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
Safety and effectiveness of ENHERTU have not been established in pediatric patients.
8.5 Geriatric Use
Of the 234 patients with HER2-positive breast cancer treated with ENHERTU 5.4 mg/kg, 26% were 65 years or older and 5% were 75 years or older. No overall differences in efficacy were observed between patients ≥65 years of age compared to younger patients. There was a higher incidence of Grade 3-4 adverse reactions observed in patients aged 65 years or older (53%) as compared to younger patients (42%).
8.6 Renal Impairment
No dose adjustment of ENHERTU is required in patients with mild (creatinine clearance (CLcr) ≥60 and <90 mL/min) or moderate (CLcr ≥30 and <60 mL/min) renal impairment [see Clinical Pharmacology (12.3)]. No data are available in patients with severe renal impairment.
8.7 Hepatic Impairment
No dose adjustment of ENHERTU is required in patients with mild (total bilirubin ≤ULN and any AST >ULN or total bilirubin >1 to 1.5 times ULN and any AST) or moderate (total bilirubin >1.5 to 3 times ULN and any AST) hepatic impairment. In patients with moderate hepatic impairment, due to potentially increased exposure, closely monitor for increased toxicities related to the topoisomerase inhibitor, DXd [see Dosage and Administration (2.2)]. No data are available in patients with severe (total bilirubin >3 to 10 times ULN and any AST) hepatic impairment [see Clinical Pharmacology (12.3)].
11 DESCRIPTION
Fam-trastuzumab deruxtecan-nxki is a HER2-directed antibody and topoisomerase inhibitor conjugate. Fam-trastuzumab deruxtecan-nxki is an antibody-drug conjugate (ADC) composed of three components: 1) a humanized anti-HER2 IgG1 monoclonal antibody (mAb), covalently linked to 2) a topoisomerase inhibitor, via 3) a tetrapeptide-based cleavable linker. Deruxtecan is composed of a protease-cleavable maleimide tetrapeptide linker and the topoisomerase inhibitor, DXd, which is an exatecan derivative.
The antibody is produced in Chinese hamster ovary cells by recombinant DNA technology, and the topoisomerase inhibitor and linker are produced by chemical synthesis. Approximately 8 molecules of deruxtecan are attached to each antibody molecule. Fam-trastuzumab deruxtecan-nxki has the following structure:
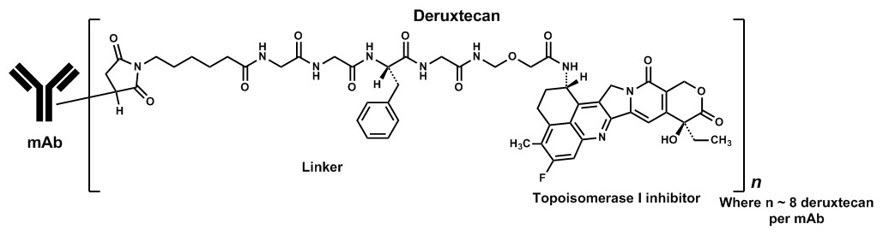
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a sterile, white to yellowish white, preservative-free lyophilized powder in single-dose vials. Each vial delivers 100 mg of fam-trastuzumab deruxtecan-nxki, L-histidine (4.45 mg), L-histidine hydrochloride monohydrate (20.2 mg), polysorbate 80 (1.5 mg), and sucrose (450 mg). Following reconstitution with 5 mL of Sterile Water for Injection, USP, the resulting concentration of fam-trastuzumab deruxtecan-nxki is 20 mg/mL with a pH of 5.5. The resulting solution is administered by intravenous infusion following dilution.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Fam-trastuzumab deruxtecan-nxki is a HER2-directed antibody-drug conjugate. The antibody is a humanized anti-HER2 IgG1. The small molecule DXd, is a topoisomerase I inhibitor attached to the antibody by a cleavable linker. Following binding to HER2 on tumor cells, fam-trastuzumab deruxtecan-nxki undergoes internalization and intracellular linker cleavage by lysosomal enzymes. Upon release, the membrane-permeable DXd causes DNA damage and apoptotic cell death.
12.2 Pharmacodynamics
Cardiac Electrophysiology
The administration of multiple doses of ENHERTU (6.4 mg/kg every 3 weeks, which is 1.2 times the recommended dosage) did not show large mean effect (i.e. >20 ms) on the QTc interval in an open label, single-arm study in 51 patients with HER2-expressing metastatic breast cancer.
12.3 Pharmacokinetics
The pharmacokinetics of fam-trastuzumab deruxtecan-nxki was evaluated in patients with cancer. Following a single dose, exposures (Cmax and AUC) of fam-trastuzumab deruxtecan-nxki and released topoisomerase inhibitor (DXd) increased proportionally over a dose range of 3.2 mg/kg to 8 mg/kg (approximately 0.6 to 1.5 times the recommended dose). At the recommended dosage of ENHERTU, the geometric mean (coefficient of variation [CV]%) Cmax of fam-trastuzumab deruxtecan-nxki and DXd were 122 µg/mL (20%) and 4.4 ng/mL (40%), respectively, and the AUC of fam-trastuzumab deruxtecan-nxki and DXd were 735 µg∙day/mL (31%) and 28 ng∙day/mL (38%), respectively, based on population pharmacokinetic analysis. Accumulation of fam-trastuzumab deruxtecan-nxki was approximately 35% at steady state (Cycle 3).
Distribution
Based on population pharmacokinetic analysis, the estimated volume of distribution of the central compartment (Vc) of fam-trastuzumab deruxtecan-nxki was 2.77 L.
For humans, DXd plasma protein binding is approximately 97% and the blood to plasma ratio is approximately 0.6, in vitro.
Elimination
The median elimination half-life (t1/2) of fam-trastuzumab deruxtecan-nxki was approximately 5.7 days. Based on population pharmacokinetic analysis, the estimated systemic clearance of fam-trastuzumab deruxtecan-nxki was 0.42 L/day.
The median apparent elimination half-life (t1/2) of DXd was approximately 5.8 days. Based on population pharmacokinetic analysis, the estimated apparent systemic clearance of DXd was 19.2 L/h.
Metabolism
The humanized HER2 IgG1 monoclonal antibody is expected to be degraded into small peptides and amino acids via catabolic pathways in the same manner as endogenous IgG.
In vitro, DXd is primarily metabolized by CYP3A4.
Specific Populations
No clinically significant differences in the pharmacokinetics of fam-trastuzumab deruxtecan-nxki or DXd were observed for age (23-96 years), race (Asian [n=291] and non-Asian [n=221]), sex, body weight (34.6-125.4 kg), mild (total bilirubin ≤ULN and any AST >ULN or total bilirubin >1 to 1.5 times ULN and any AST, n=215) hepatic impairment, mild (creatinine clearance [CLcr] ≥60 and <90 mL/min, n=206) or moderate (CLcr ≥30 and <60 mL/min; n=58) renal impairment based on population pharmacokinetic analysis.
The pharmacokinetics of fam-trastuzumab deruxtecan-nxki or DXd in patients with moderate to severe hepatic impairment (total bilirubin >1.5 ULN with any AST) or severe renal impairment (CLcr <30 mL/min) is unknown.
Drug Interaction Studies
Clinical Studies
Effect of CYP3A Inhibitors on DXd: Coadministration of itraconazole, a strong CYP3A inhibitor, with multiple doses of ENHERTU increased steady state AUC0-17 days of fam-trastuzumab deruxtecan-nxki by 11% and DXd by 18%. The impact of these changes is not clinically meaningful.
Effect of OATP Inhibitors on DXd: Coadministration of ritonavir, a dual inhibitor of OATP1B/CYP3A, with multiple doses of ENHERTU increased steady state AUC0-17 days of fam-trastuzumab deruxtecan-nxki by 19% and DXd by 22%. The impact of these changes is not clinically meaningful.
In Vitro Studies
Effects of DXd on CYP Enzymes: DXd does not inhibit CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6 and CYP3A nor induce CYP1A2, CYP2B6, or CYP3A.
Effects of DXd on Transporters: At clinically relevant concentrations (steady-state Cmax of ~0.2 µmol/L), DXd has a low potential to inhibit OAT1 (IC50 value of 12.7 µmol/L), OAT3, OCT1, OCT2, OATP1B1 (IC50 value of 14.4 µmol/L), OATP1B3, MATE1, MATE2-K, P-gp, BCRP, or BSEP transporters.
Effects of Other Drugs on DXd: DXd is a substrate of OATP1B1, OATP1B3, MATE2-K, P-gp, MRP1 and BCRP.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with fam-trastuzumab deruxtecan-nxki.
The topoisomerase inhibitor component of fam-trastuzumab deruxtecan-nxki, DXd, was clastogenic in both an in vivo rat bone marrow micronucleus assay and an in vitro Chinese hamster lung chromosome aberration assay and was not mutagenic in an in vitro bacterial reverse mutation assay.
Fertility studies have not been conducted with fam-trastuzumab deruxtecan-nxki. In a six-week repeat-dose toxicity study in rats, intravenous administration of fam-trastuzumab deruxtecan-nxki resulted in spermatid retention at 20 mg/kg and 60 mg/kg (approximately 4 and 9 times the human recommended dose of 5.4 mg/kg based on AUC, respectively). Decreased testes and epididymides weights, tubular atrophy/degeneration in testes, and reduced sperm count in epididymides were observed at a dose of 197 mg/kg (19 times the human recommended dose of 5.4 mg/kg based on AUC). In a three-month repeat-dose toxicity study in monkeys, intravenous administration of fam-trastuzumab deruxtecan-nxki resulted in decreased numbers of round spermatids in the testes at seminiferous tubule stages V to VI at ≥30 mg/kg (≥7 times the human recommended dose of 5.4 mg/kg based on AUC). Evidence of reversibility was observed in monkeys by the end of a three-month recovery period.
14 CLINICAL STUDIES
14.1 Metastatic Breast Cancer
The efficacy of ENHERTU was evaluated in study DESTINY-Breast01 (NCT03248492), a multicenter, single-arm, trial that enrolled 184 female patients with HER2-positive, unresectable and/or metastatic breast cancer who had received two or more prior anti-HER2 therapies. Patients were excluded for a history of treated ILD or current ILD at screening. Patients were also excluded for history of clinically significant cardiac disease, active brain metastases, and ECOG performance status >1. HER2 expression was based on archival tissue tested at a central laboratory prior to enrollment with HER2 positivity defined as HER2 IHC 3+ or ISH positive.
Patients received ENHERTU 5.4 mg/kg by intravenous infusion every 3 weeks until unacceptable toxicity or disease progression. Tumor imaging was obtained every 6 weeks and CT/MRI of the brain was mandatory for patients with brain metastases at baseline. The major efficacy outcomes were confirmed objective response rate (ORR) assessed by independent central review using RECIST v1.1 and duration of response (DOR).
The median age was 55 years (range: 28-96); 76% of patients were < 65 years. All 184 patients were female, and the majority were White (55%) or Asian (38%). Patients had an ECOG performance status of 0 (55%) or 1 (44%) at baseline. Ninety-two percent had visceral disease, 29% had bone metastases, and 13% had brain metastases. Fifty-three percent were hormone receptor positive. Sum of diameters of target lesions were < 5 cm in 42%, and ≥ 5 cm in 50% (not evaluable by central review in 8% of patients).
The median number of prior cancer regimens in the locally advanced/metastatic setting was 5 (range: 2-17).
All patients received prior trastuzumab, ado-trastuzumab emtansine, and 66% had prior pertuzumab.
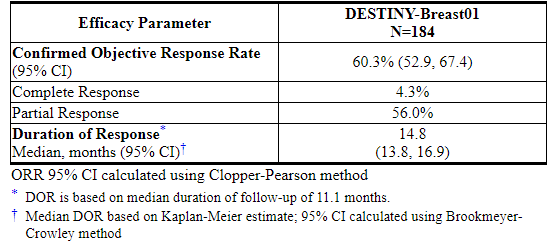
Efficacy results are summarized in Table 5.
Table 5: Efficacy Results by Independent Central Review in DESTINY-Breast01
15 REFERENCES
1.OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied/Storage
ENHERTU (fam-trastuzumab deruxtecan-nxki) for injection is a white to yellowish white lyophilized powder supplied as:

Store vials in a refrigerator at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light until time of reconstitution. Do not freeze. Do not shake the reconstituted or diluted solution [see Dosage and Administration (2.3)].
16.2 Special Handling
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a cytotoxic drug. Follow applicable special handling and disposal procedures.1
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Interstitial Lung Disease
- Inform patients of the risks of severe or fatal ILD. Advise patients to contact their healthcare provider immediately for any of the following: cough, shortness of breath, fever, or other new or worsening respiratory symptoms [see Warnings and Precautions (5.1)].
Neutropenia
- Advise patients of the possibility of developing neutropenia and to immediately contact their healthcare provider should they develop a fever, particularly in association with any signs of infection [see Warnings and Precautions (5.2)].
Left Ventricular Dysfunction
- Advise patients to contact their healthcare provider immediately for any of the following: new onset or worsening shortness of breath, cough, fatigue, swelling of ankles/legs, palpitations, sudden weight gain, dizziness, loss of consciousness [see Warnings and Precautions (5.3)].
Embryo-Fetal Toxicity
- Inform female patients of the potential risk to a fetus. Advise female patients to contact their healthcare provider of a known or suspected pregnancy [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)].
- Advise females of reproductive potential to use effective contraception during treatment with ENHERTU and for at least 7 months after the last dose [see Use in Specific Populations (8.3)].
- Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ENHERTU and for at least 4 months after the last dose [see Use in Specific Populations (8.3)].
Lactation
- Advise women not to breastfeed during treatment and for 7 months after the last dose of ENHERTU [see Use in Specific Populations (8.2)].
Infertility
- Advise males of reproductive potential that ENHERTU may impair fertility [see Use in Specific Populations (8.3)].
Manufactured by:
Daiichi Sankyo, Inc., Basking Ridge, NJ 07920U.S. License No. 2128
Marketed by:
Daiichi Sankyo, Inc., Basking Ridge, NJ 07920 and AstraZeneca Pharmaceuticals LP, Wilmington, DE 19850ENHERTU® is a registered trademark of Daiichi Sankyo Company, Ltd.
© 2019 Daiichi Sankyo Co., Ltd.USPI-ENH-C1-1219-r001



PRINCIPAL DISPLAY PANEL - 100 mg Vial Carton
NDC 65597-406-01
Rx onlyENHERTU®
(fam-trastuzumab deruxtecan-nxki)For Injection
100 mg per vial
For Intravenous Infusion Only
Dispense the enclosed Medication Guide to each patient.
Reconstitute and Dilute prior to administration
Single-Dose Vial
Discard Unused PortionCAUTION: Cytotoxic Agent
KEEP REFRIGERATED
1 vial
Daiichi-Sankyo
AstraZeneca
【备注】以上内容仅供参考,不作为用药依据,详情请参照药品附带说明书。
-
-
搜索更多相关资讯,请进入资讯页面




